Your gut is running your life. And you don't even know it.
You finally got the morning routine dialed. Eight hours on the Eight Sleep. The lift programmed by someone who actually knows what they're doing. The supplement stack. The cold plunge. Your wife says you've never looked better.
Then you walk into the gym Tuesday morning after a week of dinners and a flight back from Austin, and the bar feels like it weighs twenty pounds more than it should. You're foggy through your first call. You realize you've had three head colds this year and the last one lingered for two weeks. Your jaw is breaking out at 41 like you're seventeen. None of it is bad enough to take to a doctor. All of it is bad enough to know something underneath isn't working.
The other version of this story is on the mats. Week three of camp and the warm-up feels like quicksand. There's a circle of irritation behind your ear that wasn't there yesterday and your training partner is asking if you've checked it. The weight cut starts Friday and you already feel hollow. You haven't gotten sick this camp yet, but you got sick the last two. Your gas tank dies in round three even though you've been doing the conditioning. You're not sure why and your coach isn't sure either.
Different lives. Same organ failing both of you.
About two pounds of bacteria. Most of it lives in your large intestine. Together it runs roughly 70% of your immune system, produces most of the raw materials your brain uses to make mood and focus chemicals, decides how well you recover between sessions, and shows itself on your face when it's not working. People call it the gut microbiome.
Here's the part most of the gut-health crowd skips. Your training is already feeding it. People with better cardio fitness have more diverse gut bacteria, and the link holds up even after researchers account for diet. Strength training in previously sedentary adults grows more of the good bugs and tightens the gut lining (the thin wall between your gut and your bloodstream). The gym is part of the protocol whether you knew it or not.
But it's only one input. The rest of how you live, the dinners, the flights, the antibiotics, the daily ibuprofen for tightness, the weight cuts, the chronic stress, the convenience eating between meetings or between hard sessions, all of it pushes the gut the other way much harder than the lift pushes it back. You can be training hard and still losing this fight.
It's the operating system everything else has been running on top of.
The organ no one trained
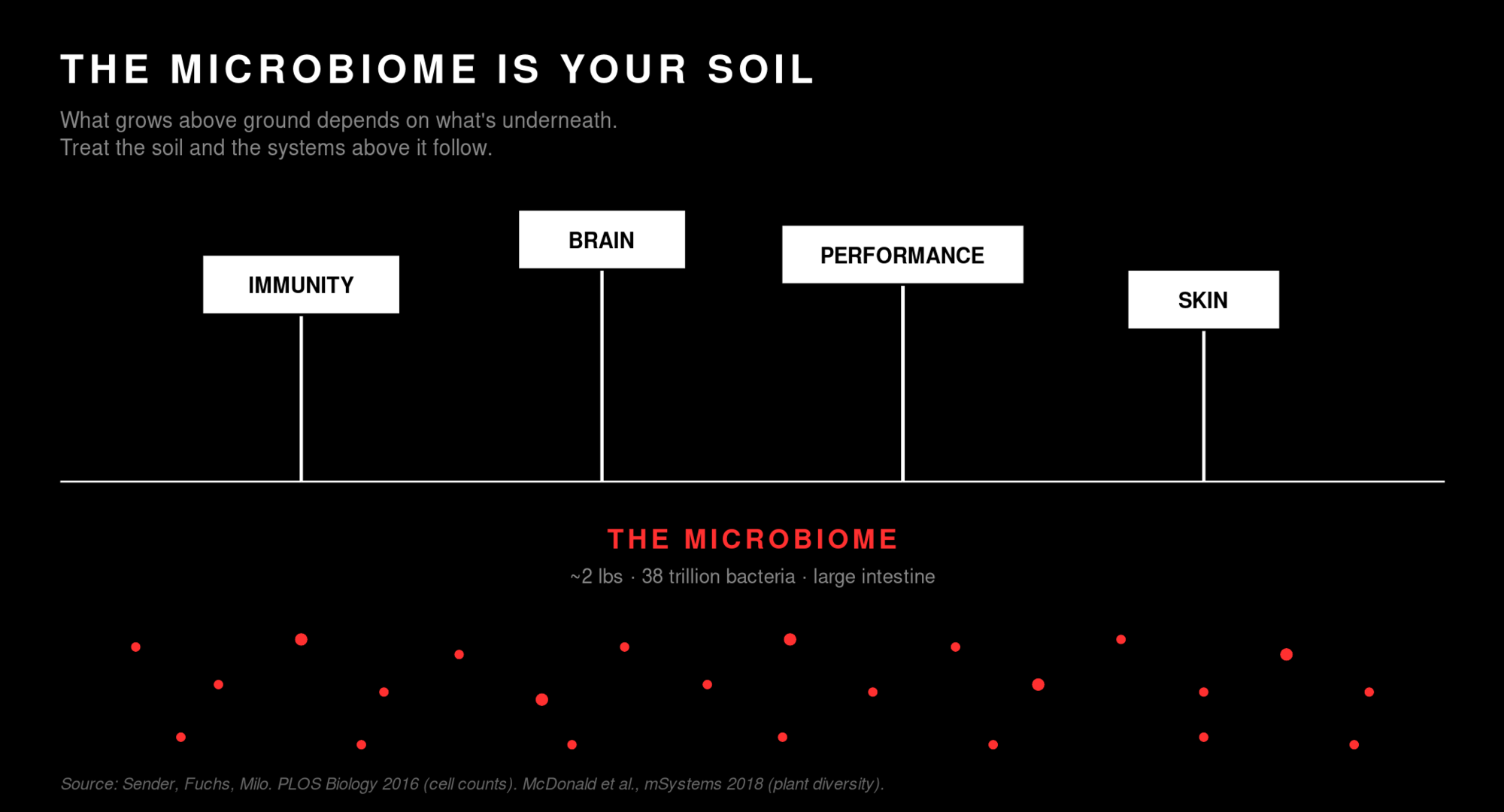
Think of the microbiome like soil.
A farm doesn't grow because of what's above the ground. It grows because of what's underneath: a living, working community of bacteria, fungi, and other microbes that decide whether a seed turns into a tomato or a stunted plant. Take that soil away, give the plants the best fertilizer you can buy, and you still don't get a harvest. The soil is doing the actual work.
The microbiome works the same way. Roughly two pounds of bacteria, most of it packed into your large intestine, with a population of about 38 trillion cells. For comparison, you have about 30 trillion of your own cells. By that count, you're slightly more bacteria than human.
Those bacteria don't just sit there. They:
Train your immune system to know friend from foe. About 70% of your immune cells are stationed right behind the gut lining, learning from what walks by.
Break down fibers you couldn't digest on your own, turning them into molecules your gut wall actually feeds on.
Talk to your brain through a thick nerve called the vagus nerve. The main phone line between your gut and your head.
Make the raw materials your body uses to build mood chemicals like serotonin (your calm-and-content chemical) and dopamine (your drive-and-reward chemical).
Influence how much fat you store, how well you absorb protein, how steady your blood sugar runs, and how inflamed your body is overall.
When the soil is good, the rest of the farm runs. When it's bad, no amount of fertilizer above ground compensates.
There are three ways the soil goes bad.
Diversity collapses. Healthy soil has hundreds of species of microbes. A monoculture is fragile. The same is true of your gut. When the variety of species shrinks (low-fiber diet, antibiotics, alcohol, chronic stress, and so on), the system loses redundancy. One bad day and the whole ecosystem tips.
The wrong species take over. Even with the same number of bacteria, the makeup matters. Some species build short-chain fatty acids (SCFAs), the molecules your gut lining feeds on. They're the most important molecules in this whole story. Others make compounds that irritate the gut and inflame the body. When the inflammatory species outnumber the beneficial ones, you have what doctors call dysbiosis. You can just call it a bad gut.
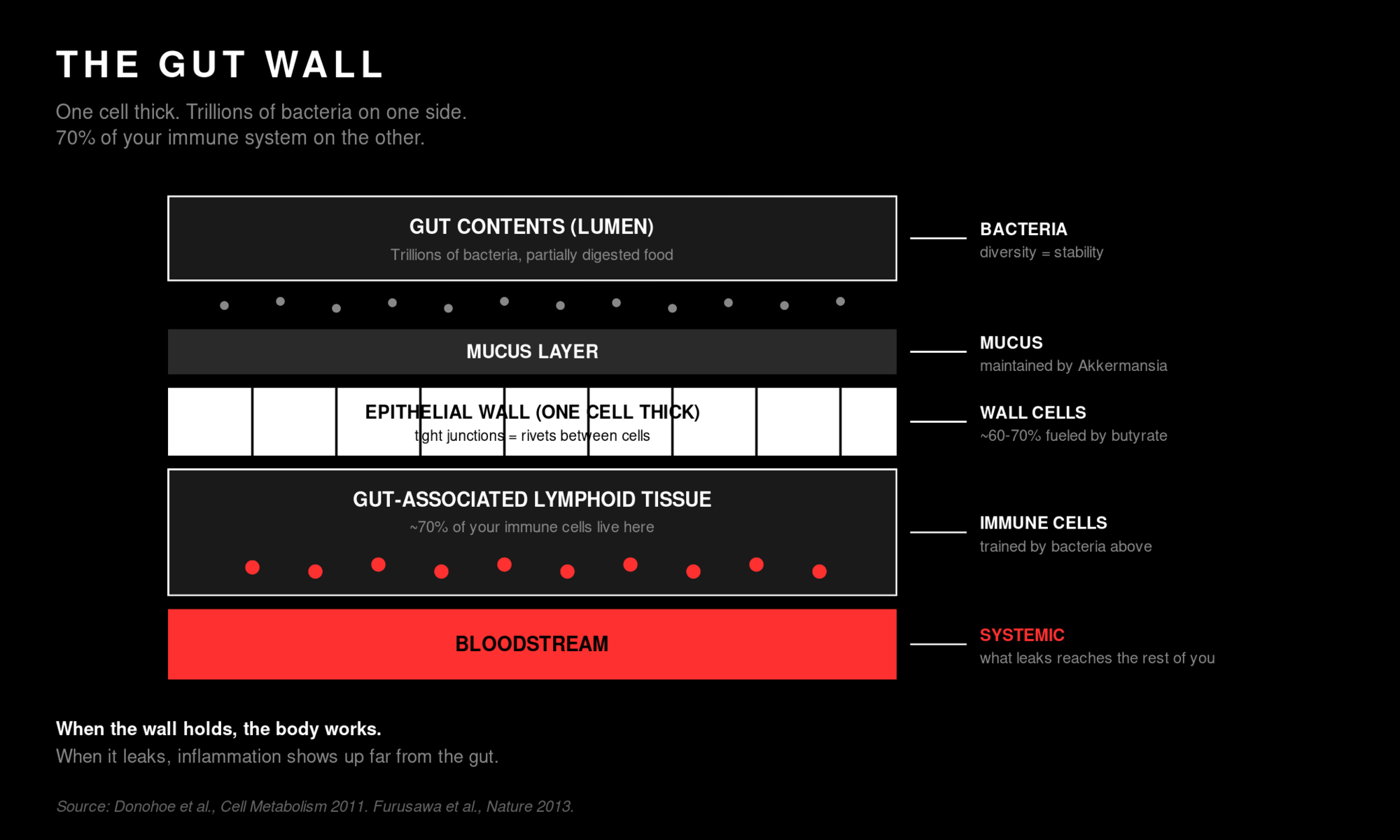
The wall thins. The gut lining itself is one cell thick. One single cell standing between your gut contents (partially digested food and trillions of bacteria) and your bloodstream. That cell layer sits under a coat of mucus that traps pathogens and feeds the good bacteria. When the bacteria above the mucus thin out, the mucus thins. When the mucus thins, the wall is exposed. When the wall is exposed long enough, the seams between cells loosen, and stuff that should stay in the gut starts leaking into the bloodstream. This is what people mean by "leaky gut."
That leak is not just a digestion issue. It's an inflammation event. Bacterial fragments end up in places they shouldn't, your immune system reacts, and the same reaction shows up far from the gut. In your joints. In your brain. In your skin. On the morning lift.
This is why the gut isn't a single-issue story. The soil sits underneath everything you're trying to grow. The rest of this piece walks through what that means for your immune system, your head, your performance, your skin, and what specifically wrecks the soil and rebuilds it.
The immune system you don't know is living in your gut
If 70% of your immune cells are right there in your gut wall, the gut microbiome isn't just connected to immunity. It is most of it.
Picture the gut wall again. One cell thick. Trillions of bacteria above it. Below it, packed into thick sheets of lymphoid tissue (the lymph nodes are part of this, but the gut version is much bigger), sits the largest population of immune cells in your body. They sit there, day after day, watching what walks by. Some bacteria they greet. Some they tolerate. Some they kill on contact. Every meal is a million-bug pop quiz, and your gut bacteria are the ones writing the questions.
When the bacteria above the wall are diverse and well-fed, the immune cells below the wall get trained accurately. They learn what to ignore, what to attack, and how hard to attack. They develop calibration. When the bacteria are sparse or wrong, the immune cells get poorly trained. They overreact to things they shouldn't (allergy, food sensitivity, autoimmune flares) and underreact to things they should be killing (viruses, opportunistic bacteria, fungi).
The molecule doing most of the talking between the bacteria and the immune cells is called butyrate. Pronounced byoo-ter-ate. Don't worry about the name. Worry about what it does. Butyrate is a short-chain fatty acid. A molecule your bacteria make when they ferment fiber from your food. It's the primary fuel for the cells of your gut lining (those one-cell-thick wall cells eat butyrate directly), and it's also a signal that tells the immune cells stationed behind the wall to stay calm. Less butyrate, hotter immune system. More butyrate, better-regulated immune system.
Get one level deeper. Butyrate does four jobs in this system.
One. It feeds the wall. The cells of your colon (the last part of your large intestine, where most of the bacterial action happens) are unusual. They get most of their energy directly from butyrate, not from glucose like the rest of your body. Estimates put this somewhere between 60 and 70% of their fuel. No fiber, no butyrate, and those wall cells start metabolizing themselves to survive. Thinner, weaker wall.
Two. It seals the seams. Between every wall cell sits a protein structure called a tight junction. Think rivets holding metal panels together on the hull of a ship. Butyrate signals those rivets to stay tight. Lose butyrate, the rivets loosen, and the wall starts leaking even when nothing else has gone wrong.
Three. It thickens the mucus. The wall sits under a coat of mucus, the first thing any bacterium or pathogen hits before it reaches a cell. Butyrate tells the cells responsible for producing that mucus to keep making more of it. Thicker mucus means more separation between the bacteria above and the wall below.
Four. It builds peacekeepers. Butyrate prompts the immune system to produce a specific type of immune cell called a regulatory T cell. These are the referees of your immune system. Their entire job is to tell the rest of the immune system to stand down when there's no real threat. Low butyrate, fewer referees, immune system on a hair trigger. This is why low-fiber diets are linked to more allergies, more autoimmune flares, and more chronic low-grade inflammation: you've lost the brakes.
Two pounds of bacteria. One molecule they make from fiber. Four jobs. Stop feeding them and all four jobs slow down at once.
If you eat fiber, you make butyrate. If you don't, you don't. You can't shortcut a butyrate level. Your bacteria have to make it from the fiber you give them, every day.
Now picture what hard training does to all of this. During heavy exercise, blood shunts away from your gut toward your working muscles. The gut lining gets less blood, less oxygen, and becomes temporarily leaky. Bacterial fragments can slip across. The immune system has to deal with the leakage at the same time it's dealing with the inflammatory load of the workout itself. For three to seventy-two hours after a hard session, parts of the immune system are tied up handling the cleanup. The classical phrase for this was the "open window" of post-exercise infection risk. The newer view is that immune cells aren't suppressed so much as redistributed (they leave the bloodstream to go patrol the linings of your gut, lungs, and skin). Either way, the practical reality stands: hard training transiently reshuffles your immune system, and a gut with bad soil makes that reshuffle much riskier.
Here's how that lands on the page for the lifter with a desk and a flight schedule.
You catch every cold the kids bring home. You used to shake them in three days, now they linger for ten. The post-flight cold has become routine. You notice you take longer to heal from small cuts than your wife does. After a few drinks at a dinner, the next day's session feels off in a way that goes beyond a hangover. The "training cold" pattern that hits you about once a quarter starts feeling like it's not random. It correlates with the weeks you slept poorly, drank more, ate worse, and trained hard anyway.
Here's how it lands in camp.
The first cold sore of camp shows up around week two. By week three you're moving slower in warm-ups. There's a scratch on your shoulder from a roll that hasn't scabbed over the way it should have. The first ringworm spot shows up the week of weigh-ins. Half your training partners get hit with something that goes through the gym in a week. You stop sleeping well during the cut. By fight night, you're not actually sick, but you're a step slower than you were six weeks ago. The cardio coach can't explain it. The S&C coach can't explain it. Both of them are right that it's not their lane.
Both stories trace to the same wall and the same molecules behind it.
There is evidence that supports plugging the gap directly. A randomized trial in marathon runners showed a 29% lower incidence of upper respiratory tract infection symptoms in the group taking a daily multi-strain probiotic (a supplement containing living beneficial bacteria) versus placebo in the week after the race. A separate trial with Lactobacillus rhamnosus GG (one of the most-studied single probiotic strains) showed that in the two weeks after a marathon, the duration of a GI symptom episode was 57% shorter in the probiotic group than placebo (1.0 day versus 2.3 days), though the difference during the months of training leading up to the race was not significant. Multiple meta-analyses now show probiotics modestly reduce the incidence and severity of respiratory infections in adults under physical stress.
That's a real effect. But probiotics are a patch on a bigger problem. What actually changes the underlying soil is everything else: fiber diversity, sleep, less alcohol, fewer antibiotic courses, and the training you're already doing. The probiotics help. They don't carry the load.
The brain you didn't know was running on gut chemistry
Your brain runs on chemicals. You already know this. What you might not know is that most of the raw materials your brain uses to make those chemicals come out of your gut, and the conversation between the two organs is constant.
Here's the cable. Running from the base of your brainstem down through your chest and into the wall of your gut is a nerve called the vagus nerve. The word "vagus" comes from the Latin for "wandering," which is accurate. It branches everywhere, but its biggest job is two-way communication between your gut and your brain. About 80% of the fibers in the vagus nerve are sensory: they're sending information up from the gut to the brain, not the other way around. Your gut is texting your brain more often than your brain is texting your gut. That gut feeling you get isn't a metaphor. It's a real signal traveling up a real wire.
Now the chemistry. Your brain doesn't just receive messages on this cable. It also depends on the gut to make the raw materials for its own mood and focus chemicals.
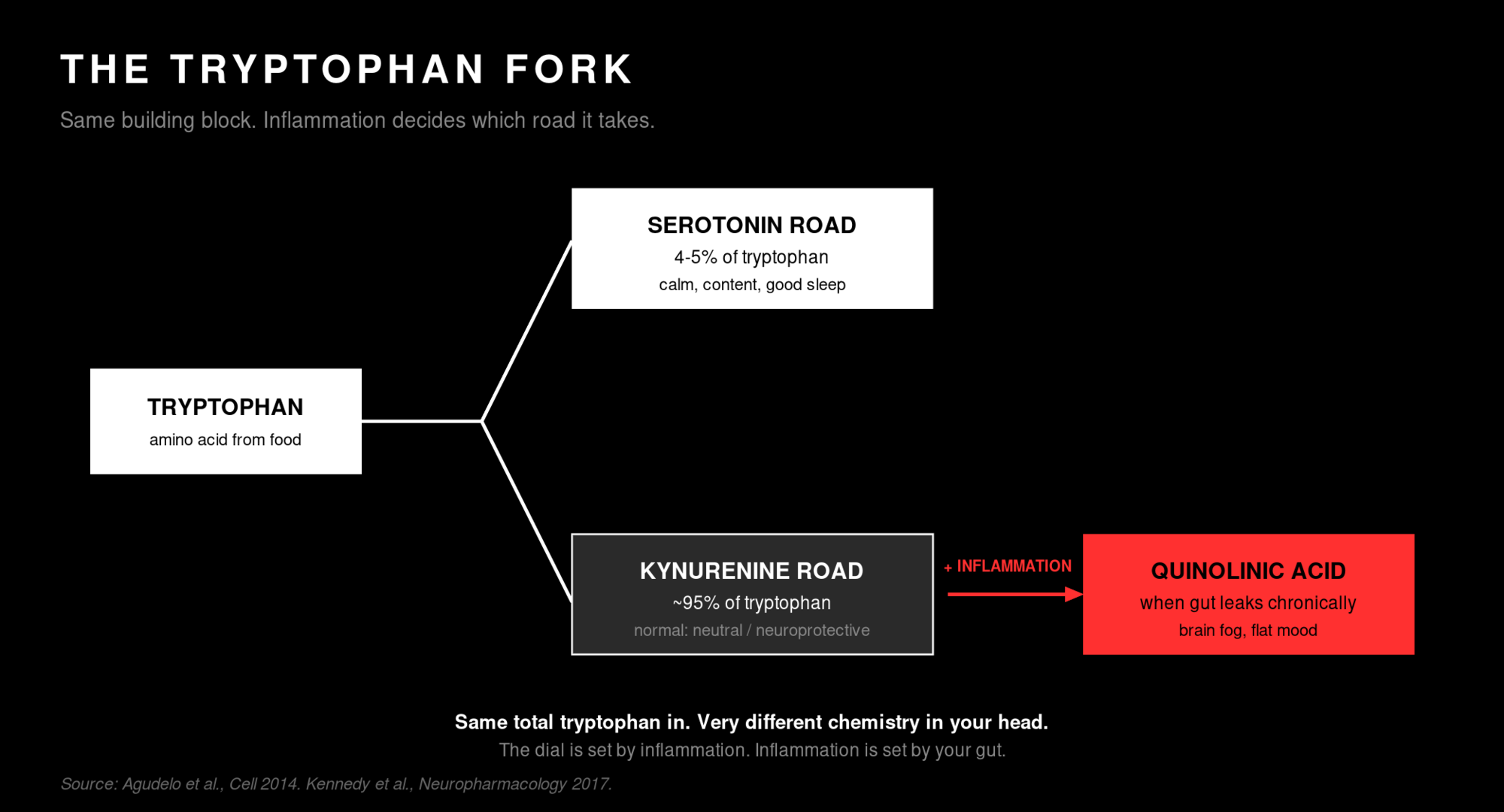
Take serotonin, the chemical most responsible for the calm-and-content state. To build serotonin, your body needs a building block called tryptophan, which is an amino acid you get from food (turkey, eggs, oats, dairy). Tryptophan crosses into the bloodstream and faces a fork in the road. Down one road, it becomes serotonin. Down the other, it becomes a compound called kynurenine.
In a healthy person, only about 4-5% of tryptophan takes the serotonin road. The other 95% takes the kynurenine road. That's not bad. Kynurenine is the precursor to other useful compounds, including ones your exercising muscles convert into neuroprotective molecules. That's a real perk of training.
But under chronic inflammation, the kynurenine road takes a darker turn. It produces compounds, including one called quinolinic acid, that are neurotoxic at high levels. They blur cognition. They flatten mood. They contribute to the kind of brain fog and low affect that doesn't respond to caffeine.
And what tips tryptophan toward the darker version of road two? Inflammation. Specifically, the low-grade, chronic, systemic inflammation that follows a leaky gut. The same leak we covered earlier isn't only an immune problem. It's a mood problem. Bacterial fragments that slip across a thinning gut wall trigger an immune response that releases inflammatory signals (called cytokines) into the bloodstream. Those signals reach the brain and switch the tryptophan dial toward kynurenine. Same total tryptophan, very different chemistry showing up in your head.
There's a second wire in this conversation. The stress system. When your brain detects threat, it activates a circuit called the HPA axis (the hypothalamus, pituitary gland, and adrenal glands, working as a chain). The end product is cortisol, the main stress hormone. Cortisol does useful things in short bursts (alertness, energy mobilization). What it does when it stays elevated for weeks and months is reshape your gut. It changes the speed of digestion, weakens the gut barrier, and shifts the bacterial population toward inflammatory species. Now you have a feedback loop. Stress causes a worse gut. A worse gut produces more inflammation. More inflammation drives more kynurenine and less serotonin. Lower serotonin reduces stress resilience. The body is then primed to perceive more threat. Loop closes. Loop tightens.
There's a third channel worth knowing. Butyrate, the molecule we covered earlier, doesn't only act on the gut. Some of it crosses from your bloodstream into your brain through what's called the blood-brain barrier (the wall that protects your brain from most things in your blood). Inside the brain, butyrate quiets inflammatory cells called microglia, supports new neural connections, and helps the blood-brain barrier itself stay sealed. A well-fed gut microbiome is, very literally, helping your brain stay calm and well-built.
Here's how that lands on the page for the lifter at the desk.
The week of three back-to-back dinners with wine, two flights, and four hours of sleep on Thursday turns into the Monday morning where you walk into your own office and can't quite get traction on the call you've taken a hundred times before. Your wife says you've been short with the kids since Saturday. You're not depressed. You're not anxious in any clinical sense. You're just running with the wrong chemicals in your head, and you can feel it as a slow tax on judgment, patience, and grip. The lift Tuesday morning feels like a chore, not a release. There's no programming explanation. There's a kynurenine explanation, and you can't supplement your way out of it. The same dinners that scrambled your gut are scrambling your mood three days later.
Here's how it lands in camp.
Week four of camp and the whole edge has dulled. You're moving well on the mats but your training partner is hitting things you'd usually see coming. You're irritable in ways you can't fully account for. Sleep gets choppy. The morning weigh-ins on the home scale start producing a flat dread you don't remember feeling in your last camp. By weigh-in day itself, you're a different person than you were six weeks earlier. Some of that is dehydration and food restriction. Some of it is real. Underneath, a hard cut combined with weeks of training inflammation is dragging the kynurenine dial all the way over. Less serotonin made. More neurotoxic byproducts in circulation. Your gas tank is one piece of the story. Your head is another. The technical work that needs the clearest version of you is happening at the bottom of the well.
Both of these are not a willpower problem. They're a gut problem showing up on the wrong floor of the building.
A note on supplements before we move on. There are randomized trials testing what researchers call "psychobiotics," which is a marketing-adjacent term for probiotics studied for mood effects. Some show real reductions in anxiety, stress, and cortisol (a study with Lactobacillus plantarum showed clear drops at 8 weeks in stressed adults). Others show modest effects only in people whose lifestyle baseline is already good. Strain matters. Population matters. The fix isn't a pill. The fix is the soil. Fiber, fermented foods, sleep, light alcohol, fewer antibiotic courses, and time outside all change the underlying gut more than any capsule will.
The brain is downstream of the gut. Treat the soil, and the chemistry above it changes.
The gut behind your gas tank
If you've ever wondered why your conditioning doesn't match your output, why the third round dies even though your interval work has been good, or why the deadlift you hit clean three weeks ago is suddenly grinding, this section is the one for you.
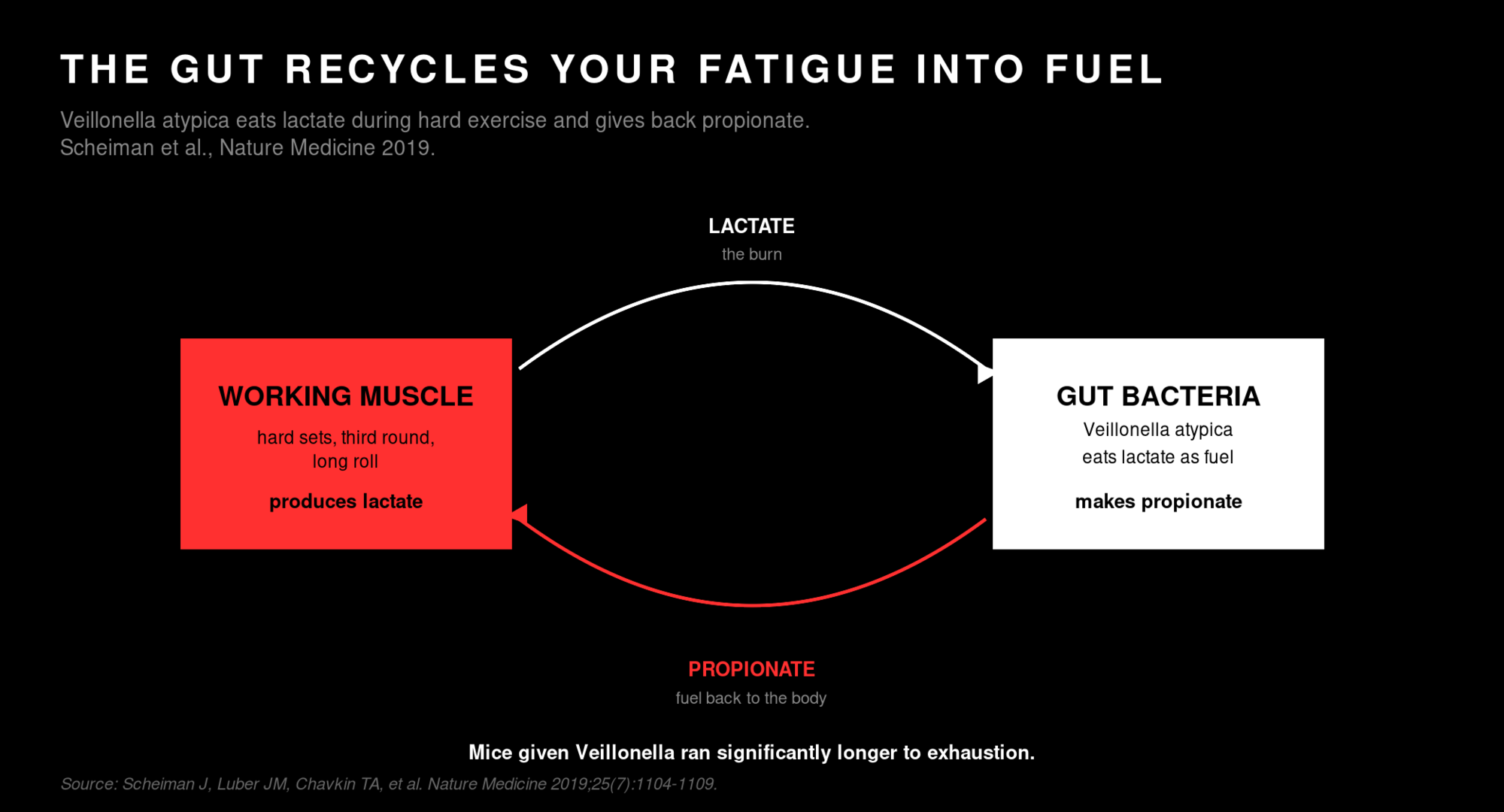
In 2019, a group of researchers at Harvard sampled stool from Boston Marathon runners before and after the race. They found that one specific bacterium spiked in abundance after the marathon. They named it Veillonella atypica. Then they did the thing scientists do when they're suspicious of an interesting finding: they isolated the strain, grew it up, and put it into mice. The mice ran significantly longer to exhaustion on a treadmill than control mice did. Then they figured out the mechanism.
Veillonella eats lactate. Specifically, it eats the lactate your muscles produce during hard exercise. The very stuff that's been blamed for "the burn" for decades. What Veillonella does with the lactate is turn it into a short-chain fatty acid called propionate. Propionate then circulates back into your system and acts as fuel. To finish proving it, the researchers injected propionate directly into mice and reproduced the same endurance boost. There's a bacterium in your gut whose job is essentially to recycle your fatigue back into energy.
You don't have to memorize the name. Just understand the implication. Endurance, recovery, and how well you tolerate hard work are not just questions of cardio capacity and protein intake. They're questions of who lives in your gut and what they do with what you make during a workout.
The story repeats across other species. Endurance athletes who train more than sixteen hours a week have unusually high levels of a bacterium called Prevotella in their guts. Prevotella is good at processing complex carbohydrates, which is what an endurance athlete needs. The harder they train, the more of it they have. Resistance training in sedentary adults shifts the population toward more butyrate-producing species, which lowers inflammation and tightens the gut wall. The gut microbiome rearranges itself around what you do, and it gets noticeably better at supporting hard work over time.
Now consider what wrecks this on the other side. Inflammation.
When you train hard, you produce inflammation on purpose. That's part of the signal that makes you adapt. The body is supposed to clean it up quickly between sessions. If your baseline inflammation is already running high because your gut is a mess, you don't clean it up. You just stack workouts on top of unresolved inflammation. Muscle soreness lingers. Sleep gets choppy. Soft tissue takes longer to remodel. The work you did yesterday is still costing you on the lift today.
There's a tell here that's worth naming. Most lifters and fighters who hit a stuck point reach for ibuprofen or naproxen for soreness and chronic tightness. The data on that is not flattering. Studies on endurance athletes show that NSAIDs (the drug class of ibuprofen, naproxen, aspirin) don't reduce delayed-onset muscle soreness, and the athletes who take them before exercise show higher markers for inflammation and cell damage afterward than the athletes who don't. The painkiller is making the recovery problem worse. We'll handle this fully in the wreckers section. For now, file it.
Here's how that lands on the page for the lifter who's already doing the work.
You've been programming smart. Two main lifts a week, the right accessories, a deload every four weeks. Your sleep is decent. Your food is "fine." Then you walk in for a Tuesday top set and the bar grinds in a way that doesn't match what your sheet says you should be doing. The squat that felt crisp three weeks ago feels heavy. The same bench complex that left you fresh now leaves you sore for three days. You blame the program. The program is fine. Your recovery is what's not landing. The inflammation from last week's dinners and Friday's flight is still on the table, and you're trying to add this week's work onto a system that hasn't cleared yesterday's bill. The plateau isn't a strength problem. It's a clearance problem. And the only way to fix it is upstream of the bar.
Here's how it lands in camp.
Your conditioning is built. You've done the hill sprints, the bike intervals, the rounds of grappling drills at threshold. On paper you should be fresh into round three of your next session. In practice, the third round is the worst it's been in months. Your forearms gas out. Your timing slows. The cardio coach checks your zones and they look fine. The S&C coach checks your loads and they look fine. What's happening is that lactate is piling up faster than you can clear it because the bacterial system designed to clear it (the Veillonella story) is starved for the right inputs. Your inflammation baseline is already high from camp volume, your gut wall is thin from the cut, and the very microbes that recycle fatigue into fuel are not there in the numbers a serious training week needs.
So what actually rebuilds this part of the picture? Some of it is the same answer that runs through the rest of the piece: fiber diversity, sleep, and the training you're already doing. But a few specific foods hit recovery and performance hard enough to be worth naming here.
Kiwi. Two a day gives you about 4-6 grams of fiber (mostly the prebiotic kind that feeds gut bacteria), a heavy dose of vitamin C for connective tissue and immune repair, and a natural enzyme called actinidin that helps break down dietary protein. For an athlete eating a lot of protein, this is the small input that helps the rest of the food work harder.
Broccoli sprouts. Not mature broccoli, sprouts. These are the three- to five-day-old germinations sold as little tufts at the store, and they contain anywhere from ten to one hundred times more of a precursor compound called glucoraphanin (which the body converts to sulforaphane) than mature broccoli does. Sulforaphane activates the body's main anti-inflammatory and antioxidant pathway. A small handful a few times a week tilts the inflammatory baseline downward. Less unresolved inflammation. Faster between-session recovery.
Blended chia seeds. Whole chia is fine. Blended is better, because the blend cracks the seed and releases both the soluble fiber (the kind that ferments into short-chain fatty acids) and the polyphenols, which feed mucin-protective species like Akkermansia. A small daily spoon in a smoothie does the work.
Kefir. Worth a dedicated mention. Yogurt typically has a small number of bacterial strains (often just the two starter cultures plus a few added probiotics). Kefir has many more: typically 10 to 30 distinct bacteria and yeasts, with traditional fermentations sometimes reaching 50 or more. A single glass introduces a much wider population of beneficial species. For someone whose gut diversity is the bottleneck, kefir is a faster lever than most probiotic capsules.
Polyphenol-rich foods. Pomegranate, dark berries, green tea, extra virgin olive oil, cocoa, grapes. These feed Akkermansia muciniphila, the species that sits in the mucus layer and tells the gut to keep producing it. We'll come back to Akkermansia in the skin section because the same bug shows up there too.
None of these is dramatic on its own. The point isn't to add one item and wait for a transformation. The soil responds in days to weeks when the right inputs come in. A camp that adds kiwi, sprouts, and kefir for six weeks runs differently than a camp that doesn't. A founder's training quarter where the food gets cleaned up in this specific way recovers faster than the one before it. The change is upstream of the bar and upstream of the third round, and it isn't a supplement.
Treat the gut and the gas tank deepens.
The face that's keeping a chart of your gut
Your skin is the largest organ in your body. It's also the most visible reflection of what's happening underneath. Most of the time, when skin doesn't look right and there's no obvious surface cause, the explanation lives in the gut.
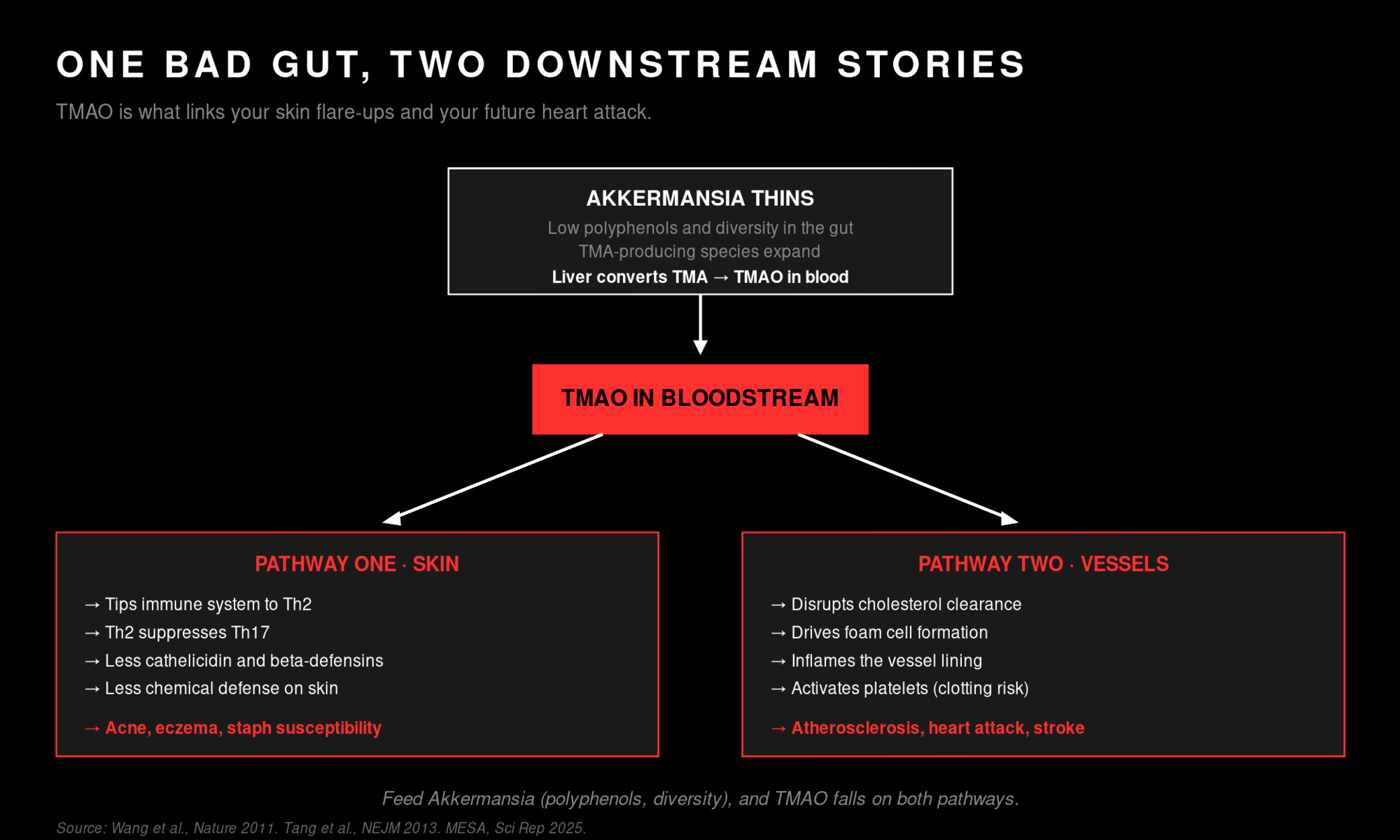
Here's the mechanism, in plain English. The mucus layer above your gut wall is maintained by a specific bacterium called Akkermansia muciniphila (the one we saw at the end of the performance section). When you eat polyphenol-rich foods like dark berries, green tea, olive oil, pomegranate, and cocoa, Akkermansia thrives, the mucus layer gets thicker, and the gut wall stays intact. When you don't, Akkermansia thins out, and other species step into the gap.
One group of those species produces a compound called TMA, which the liver converts to TMAO (trimethylamine N-oxide). When TMAO rises in the bloodstream, it tips a specific branch of the immune system toward what's called a Th2 response. That's the immune system's "allergy and inflammation" channel. More Th2 activity means more histamine, more IgE (the antibody behind most allergic reactions), and more inflammatory traffic showing up at the skin.
Multiple studies in the gut-skin axis literature now trace this exact chain. Mouse work shows that depleting Akkermansia in the gut shifts the immune profile and worsens skin inflammation. Human studies in patients with atopic dermatitis (eczema) show reduced beneficial gut species, higher inflammatory metabolites, and disease severity that tracks with these gut-derived signals. Same root cause as your gut's inflammatory cycle, surfacing on the outside.
But the Th2 skew does more than drive eczema. It also disarms your skin's chemical defense system.
The outermost cells of your skin (called keratinocytes) make a class of small molecules called antimicrobial peptides, or AMPs. AMPs are essentially the skin's natural antibiotics. They sit in the surface oils and the lower layers, and their job is to kill or weaken bacteria, fungi, and viruses before those organisms can take hold. Two of the most important ones go by the names cathelicidin (specifically a peptide called LL-37) and the beta-defensins. They are particularly good at killing Staphylococcus aureus, including the methicillin-resistant version known as MRSA.
Here's the catch. AMPs get produced most strongly in response to a Th17 immune signal. Th17 is a different branch of the immune system from Th2, and the two channels compete with each other. When the gut is in good shape, the immune system maintains a healthy Th17 response, and the skin manufactures AMPs on a normal schedule. When the gut is inflamed and TMAO is high, the system tips toward Th2, and Th2 actively suppresses Th17. The cytokines that drive Th2 (IL-4 and IL-13) interfere with Th17's signal to the skin, and AMP production drops. The chemical defense disarms.
This is why people with atopic dermatitis have measurably lower levels of cathelicidin in their skin, and why they get colonized with S. aureus far more often than people without it. It's the same mechanism, simpler version, for anyone with a chronically inflamed gut. Less AMP. More colonization. More inflammation. More flares of acne, folliculitis, slow-healing cuts, and (for the people on mats) more risk of the next staph outbreak going through the gym landing on you.
The skin has a second defense system that works alongside this one, which is the population of beneficial bacteria living on the surface itself (Staphylococcus epidermidis and its allies). They compete with S. aureus for real estate and produce their own kill compounds. We'll cover that side of the story later, in the section on combat sports skin defense. For now, the point is that your gut is responsible for half of the skin's defense system, the chemical half. When it's working, the AMP production stays up, and the everyday opportunists (the staph in the gym, the fungi in shared shoes, the cold sore that wants to come back) don't get the foothold they need.
What does that look like in real life?
The jaw breakouts we talked about at the start. Not the hormone-driven teenage kind. The ones that show up around the jawline and lower face in your thirties and forties, in the rhythm of bad weeks. They track travel, alcohol, fast food, and stretches of bad sleep. They're inflammatory acne, and the inflammation is rising up from a gut that hasn't been fed right.
Eczema flares, especially the ones around the elbows, behind the knees, on the eyelids. These often track an immune system that's gone Th2-heavy, which is the same channel that drives seasonal allergies and food sensitivities. Same dial, different face.
Rosacea, the persistent red flush on the cheeks and nose, has a documented overlap with small intestinal bacterial overgrowth (SIBO), where bacteria that shouldn't be in the small intestine colonize it and create chronic inflammation. People treated for SIBO often see their rosacea improve substantially.
Slow-healing cuts and scratches. When the gut is leaking inflammatory signals into the bloodstream, the body's repair processes get slower across the board. Small abrasions stay scabbed longer. Soft tissue takes more time. The athlete or grappler watching a roll-scratch stay open three days longer than usual is reading real data about their inside.
Dull, tired-looking skin in general. Most of the "glow" people associate with healthy skin is a function of hydration, vascular flow, and a calm immune layer underneath. All three are downstream of gut function.
The good news is that skin responds visibly when the gut work starts. Acne and rosacea typically need a few weeks. Eczema flares can settle inside one to two weeks once the inflammatory load drops. People who do nothing more dramatic than add kiwi, blended chia, fermented foods, and a half-cup of dark berries to their day, while cutting alcohol back, often see meaningful skin changes inside a month. Not from a serum. Not from a peel. From the soil.
The two-pound organ in your large intestine has a postal route to your face. When it's healthy, the route stays quiet. When it's stressed, you read the news in the mirror.
The heart and vessels your gut is shaping every day
There's one more system worth naming before we get into what's wrecking the soil. The cardiovascular system. Most people think of gut health as a digestion conversation and stop there. The reality is that the gut microbiome is doing measurable work on your blood pressure, your blood vessel lining, and your long-term risk of the most common cause of death in the developed world. It runs through two paths we've already met in this piece.
Path one: TMAO and the arteries. We covered TMAO in the skin section as the molecule that tips the immune system toward Th2 and disarms the skin's defenses. The same molecule does something else on the way through your bloodstream. It promotes the buildup of plaque in your arteries.
The story goes like this. When you eat foods high in choline, L-carnitine, or lecithin (mostly animal products like red meat, eggs, and full-fat dairy), gut bacteria in a depleted microbiome produce a precursor called TMA. The liver converts TMA into TMAO. Some TMAO is normal and not a problem. The problem is when the conversion runs high, which happens when the gut is short on Akkermansia muciniphila and other beneficial species and long on the TMA-producing ones.
TMAO drives three cardiovascular effects when it sits elevated in the blood. First, it disrupts the way your body processes cholesterol, particularly the "reverse cholesterol transport" that clears excess cholesterol out of artery walls. Second, it primes immune cells called macrophages to turn into "foam cells," which is the first cellular step in atherosclerotic plaque formation. Third, it activates platelets, making blood slightly more prone to clotting.
The clinical data is hard to ignore. A 3-year follow-up of 4,007 patients undergoing coronary angiography found that elevated plasma TMAO predicted major adverse cardiovascular events (heart attack, stroke, death) independent of standard risk factors like LDL cholesterol and blood pressure. A 2025 analysis from the Multi-Ethnic Study of Atherosclerosis confirmed graded risk across quintiles: people in the highest TMAO quintile had roughly a 33% higher hazard of an atherosclerotic cardiovascular event compared to the lowest. The honest caveat is that TMAO is not a fully proven causal factor (some of the association is mediated by kidney function), but the consistency of the data across populations is real.
The Akkermansia we covered in the skin section is the same bug whose loss opens the TMAO production line here. Polyphenol-rich foods (pomegranate, dark berries, green tea, extra virgin olive oil, cocoa) don't just feed your skin defense. They cap the cardiovascular line too.
Path two: butyrate and blood pressure. This one runs through the molecule from the immune section. Butyrate, and to a lesser extent the other short-chain fatty acids (acetate and propionate), does more than feed the gut wall and calm the immune system. It also helps regulate blood pressure.
The mechanism is mostly through specific receptors on the cells that line your blood vessels (the endothelium). Those receptors respond to SCFAs by relaxing the vessel wall. More SCFAs from fiber-fermenting bacteria, more relaxation, lower blood pressure. Less SCFAs, more vascular tension, higher pressure.
People with hypertension consistently show a different gut microbiome composition than people with normal blood pressure. Fewer SCFA-producing species (Faecalibacterium, Roseburia, the same names that keep showing up across this piece) and lower fecal SCFA levels. Animal studies that directly supplement SCFAs lower blood pressure measurably in hypertensive models. Human intervention studies feeding high-fiber diets to people with elevated blood pressure show modest but real reductions, on the order of 4-6 mmHg systolic in the better trials.
This is not a replacement for blood pressure medication if you need it. It is a real piece of the puzzle most standard cardiology visits never raise.
Here's how that lands for the lifter at the desk.
You're 41. Your last physical showed blood pressure of 132/85, which the doctor called "borderline" and told you to "watch." Your LDL came back at 142, also "borderline." You've been telling yourself you'll get to it. You eat what you think is a clean diet. You lift hard. You don't smoke. The blood pressure cuff readings keep creeping up year over year. The conversation you're not having yet is whether you'll be the same operator at 55 as you are now, or whether the cardiac event that takes most men in your demographic will take you too. The gut work in this piece is not the only lever on that question. It's a lever the standard cardiology visit will not mention.
Here's how it lands for the combat athlete.
You don't think much about cardiovascular disease yet because you're in shape. The metrics that matter more right now are the cardiac drift you feel in the third hard round, the elevated resting heart rate during a hard camp, the post-fight blood pressure that takes longer to settle than it used to. These aren't all gut-driven, but inflammation downstream of a thin gut wall feeds into all of them. The same fight-camp diet that produces an empty microbiome ecosystem produces less SCFA-driven vascular relaxation and more inflammatory tone in your vessels. Your conditioning is asking your heart to do hard work session after session, and the soil is sending it the wrong chemistry.
The relief side looks identical to the food playbook we'll consolidate later, with two emphases worth flagging. Polyphenol foods (pomegranate, dark berries, green tea, extra virgin olive oil, cocoa) earn extra weight in this conversation because they feed Akkermansia and cap TMAO production. Fiber diversity drives SCFA production, which drives vascular relaxation and lower pressure. Fermented foods do both. Less alcohol means less inflammation of the blood vessel lining and less TMAO from secondary pathways. Training itself protects you here: exercise independently lowers cardiovascular risk and increases SCFA-producing bacteria, which is part of why fit people get more cardiovascular benefit than their cholesterol numbers alone would predict.
Treat the soil and the heart and vessels above it keep showing up for you, not against you.
What's wrecking the soil for the lifter at the desk
The gut doesn't fall apart overnight. It falls apart through repetition. Most of the wreckers below are things you've done a thousand times without noticing the cost. There's no single villain. There are six or seven small ones working in concert.
These are the ones doing the most damage to a typical operator's gut, in roughly the order of how often they show up.
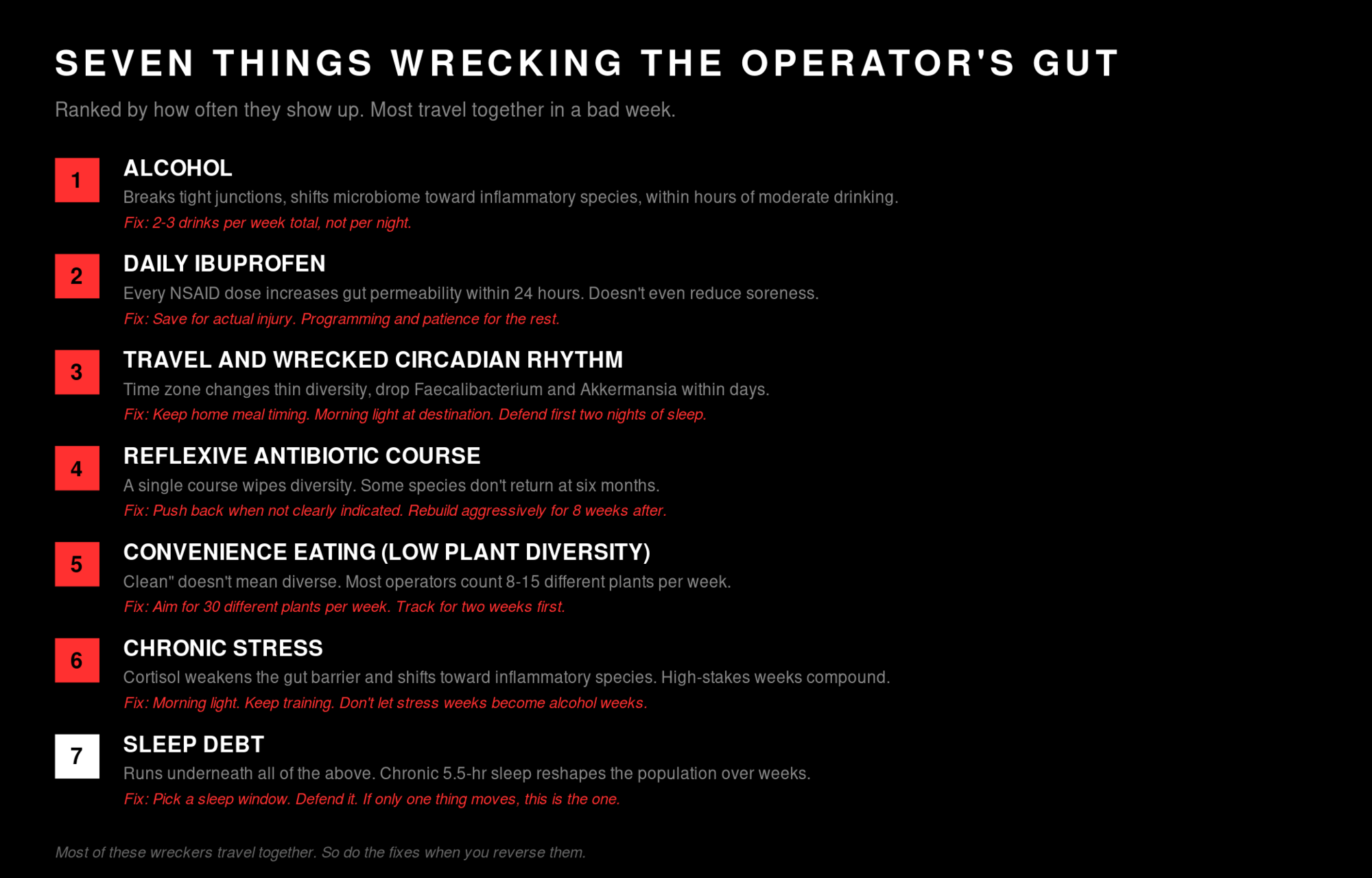
Alcohol. The two glasses of wine with dinner three or four nights a week. The cocktail at the conference reception. The bourbon Thursday with a client. None of those individual drinks feels meaningful, and any single night you'd swear it didn't bother you. The problem is what happens at the cellular level even with moderate drinking. Alcohol breaks down the protein structures (tight junctions, the rivets we covered earlier) that hold the gut wall together, increases gut permeability within hours of a few drinks, and shifts the bacterial population away from beneficial species like Lactobacillus and Bifidobacterium and toward the inflammatory ones. Bacterial fragments slip across, end up in the bloodstream, and trigger systemic inflammation. The next morning's hangover isn't only your liver. It's also your gut signaling that the wall got breached. Cut alcohol back to two or three drinks a week (not per night), and most people see meaningful gut, mood, and sleep changes inside three weeks.
Daily ibuprofen. This one is hidden in plain sight. Operators who lift hard often have chronic tightness somewhere. The shoulder. The lower back. The hip. The default becomes a Costco-sized bottle of ibuprofen or naproxen on the kitchen counter, with a few pills before training "just to take the edge off." Every NSAID dose increases gut permeability within twenty-four hours. Taken before exercise, the damage compounds: the gut is already shunting blood away from the lining during the workout, and the drug is loosening the rivets on top of that. The published data on athletes taking ibuprofen before training shows higher gut-injury markers, higher systemic inflammation, and (the kicker we covered earlier) no actual reduction in muscle soreness. The painkiller doesn't help recovery and damages the system you need for recovery. If your shoulder is barking, the fix is in programming, soft tissue work, and patience, not the pill that breaks the lining you eat through. Save NSAIDs for the actual injury, the bad sprain or the surgical recovery. Not the daily background ache.
Travel and the wrecked circadian rhythm. Founders fly. Often. The cost shows up in the gut faster than it shows up anywhere else. Your gut bacteria have their own daily clock, synced to your eating and sleeping patterns. When you fly across time zones, eat at the airport at the wrong hour, and sleep four hours in a hotel bed, that clock breaks. Diversity drops. Faecalibacterium and Akkermansia, two of the most beneficial species in a healthy gut, are particularly sensitive to circadian disruption. Within days of a hard travel week, the lining gets thinner and inflammation rises. This is why the post-flight cold is so reliable. The mitigations are unglamorous: keep meal timing on your home schedule when abroad if you can, get morning light at the destination as soon as possible, prioritize the first two nights of sleep, and add fiber and fermented foods on travel days specifically (a kiwi on the plane, a yogurt or kefir on landing).
The reflexive antibiotic course. Most operators have a story like this: a lingering sinus thing, a dental issue, a UTI, a doc-in-a-box visit that ends with a Z-Pak or amoxicillin "just to be safe." A single course of broad-spectrum antibiotics knocks out diversity dramatically, and the recovery is incomplete. Studies tracking gut composition after one antibiotic course show that some species never come back, even six months out. This doesn't mean refuse antibiotics. They save lives. It means use them when they're actually indicated, push back gently when they're not (most upper-respiratory infections are viral and antibiotics do nothing), and rebuild aggressively in the weeks after a course with fiber diversity, fermented foods, and time.
Convenience eating. This is the quiet wrecker, because no single meal feels like a big deal. The protein bar at 10am. The salad with chicken at noon. The smoothie. The takeout sushi. The protein shake. None of it is obviously bad. The problem is what's missing. The standard operator diet, even when "clean," tends to run very low on the one input the microbiome actually needs: plant diversity. A microbiome fed by ten different plants in a week is a different ecosystem from one fed by thirty. Low diversity means lower SCFA production, thinner mucus layer, weaker barrier, less butyrate, all of the chains we've walked. The fix isn't a fancier protocol. It's a counting exercise. Aim for thirty different plants a week (vegetables, fruits, legumes, nuts, seeds, whole grains, herbs and spices all count). Most operators land between eight and fifteen when they actually start counting. Tripling that number is the single highest-leverage food change you can make.
Chronic stress. The stress loop we covered earlier is one of the biggest gut-disruptors in the operator life, and it's the hardest one to address because most of your high-stress weeks are also your high-stakes weeks. Cortisol weakens the gut barrier, changes the speed of digestion, and shifts the bacterial population toward inflammatory species. The combination of a chronic-stress baseline and a low-fiber diet is particularly destructive. You're driving the loop with one hand and starving the recovery with the other. The mitigation isn't "stress less," which is useless advice for people running real businesses. It's targeted: protect sleep aggressively, get outside daily for at least twenty minutes of morning light (which dampens cortisol), keep training going (exercise is the most reliable cortisol regulator we have), and do not let the chronic-stress weeks also become the chronic-alcohol weeks.
Sleep debt. This one runs underneath all of the above. The microbiome rebuilds and shifts on a daily cycle, and that cycle is set by your sleep. Even a few nights of bad sleep start to shift gut bacterial diversity. A chronic 5.5-hour sleep pattern reshapes the population over weeks toward the species linked to inflammation and metabolic dysfunction. There is no probiotic, no diet, and no protocol that compensates for chronically inadequate sleep. If only one thing on this list moves, make it this one.
Most of these wreckers travel together. The week of the bad pitch is the week of two extra drinks, one missed gym day, four hours of sleep on Thursday, an emergency ibuprofen, and three takeout meals. The gut takes a beating from all of them at once. That's why the symptoms (Monday fog, jaw acne, the bar grinding on Tuesday, the cold that won't shake) tend to cluster. They aren't separate. They're the same week showing up on different floors of the building.
The relief side: the wreckers also tend to retreat together. The week you protect sleep, eat thirty plants, hold alcohol to one or two, skip the daily painkillers, and stay outside in the morning is the week you sleep better, lift better, think more clearly, and look better. The soil rebuilds fast when the inputs come back online. Two to four weeks of clean inputs is usually enough to feel a meaningful change. Two to three months is enough to change the underlying composition substantially.
The other side of this story is the combat athlete, whose wreckers overlap with the operator's but include a few of their own.
What's wrecking the soil for the combat athlete
Combat athletes do most of the same damage to their guts that operators do (the chronic stress, the NSAIDs for training pain, the bad sleep weeks). What makes the combat athlete's gut different is what sits on top of all that. Five specific patterns hit harder in fight camp than they do anywhere else.
Weight cuts. Between 60 and 80% of competitive combat athletes engage in some form of weight cut for competition. Even the "moderate" version is one of the most aggressive things you can do to your gut. Rapid weight loss through restriction and dehydration reduces beneficial bacterial diversity, increases intestinal permeability (the wall thinning we keep coming back to), and impairs the absorption of recovery nutrition the moment you most need it. Even mild dehydration (2-3% body mass) increases gut permeability measurably. The 5-8% body mass cuts you see most commonly in MMA and BJJ produce a real, documented hit to the gut barrier and the microbiome composition. This is why you feel hollow on cuts. The food restriction is one piece. The wall thinning, the inflammation rising, and the immune system patrolling a compromised barrier while your nervous system is also handling dehydration and low blood sugar are the rest of the story. The fix isn't to skip the cut. For most athletes, the cut is non-negotiable. The fix is to do it the way the recent sports nutrition guidelines recommend: gradual, mostly through nutrition and modest manipulation of carbohydrate and salt, with dehydration as the last lever rather than the first. The acute cuts that go from 178 to 155 in five days are the ones that wreck the gut for weeks afterward. The athletes who cut six pounds over the last week, mostly through reduced sodium and gut content, recover their training within days of weigh-in.
Repeated antibiotic exposure. Most combat athletes who've been at it for a few years have a long antibiotic history. Last year's staph infection. The ringworm course. The MRSA scare. The "let's just hit it with a course of Cipro" from the team doctor. Each round of antibiotics wipes out diversity. The recovery is partial. Some species don't come back. Now overlay this onto the gut-skin axis we covered earlier: when the gut is depleted, the immune system's calibration drifts toward Th2, the chemical defenses on your skin (the AMPs we covered) drop, and the next skin infection comes more easily. Which means another antibiotic course. Which means more disruption. Which means more skin vulnerability. The loop tightens, and most fighters don't realize they're inside it.
The first move out of the loop is rebuilding the gut hard between courses. Fiber diversity, fermented foods, kefir (the 30-50-strain version of yogurt), and aggressive sleep for at least eight weeks after any course. The second move is being precise about when antibiotics actually go. A ringworm flare is not staph. Most skin issues respond to topical treatment plus mat hygiene plus better gut work. Save the systemic antibiotics for the times when they actually save you.
The fight camp diet. Most fight camps run on the same diet template: chicken or fish, white rice, broccoli, eggs, oats, a couple of fruits, repeat. It hits the macros. It misses the microbiome. The standard fight-camp food rotation produces about eight to twelve unique plants in a week. The microbiome wants thirty. Combine this with the camp's high training volume (which increases inflammation that the gut is supposed to clean up) and you have a system where the soil is starved at exactly the moment it needs the most inputs. The reason the cuts and the camps feel harder than they should is partly this: you've been eating a clean diet that produces an empty ecosystem. The fix is small and specific. Rotate plants more aggressively. Add a kiwi a day. Add a half-cup of berries. Add a tablespoon of blended chia. Add a glass of kefir. Add three or four different herbs and spices to the same chicken. Add legumes back in (the standard restricted fight-camp diet often cuts these out unnecessarily for athletes without actual IBS). The macro numbers don't have to change. The diversity number does.
Saunas and dehydration cycles. Combat athletes use heat exposure throughout camp, not only during the cut. The daily sauna sessions. The hot baths. The cardio in the sauna suit. Heat exposure has real benefits for cardiovascular adaptation and heat acclimation. It also dehydrates you repeatedly, and repeated dehydration cycles compound the gut barrier damage that the eventual cut amplifies. The body never fully rehydrates between sessions during a hot camp. Gut permeability stays elevated. Inflammation stays elevated. This isn't an argument against heat work, which has too many upsides to abandon. It's an argument for hydrating aggressively between sauna sessions (not only sips during the session, but real volume after), keeping electrolytes balanced, and giving the gut a recovery window of normal hydration on training off-days rather than continuing the squeeze.
The intra-camp infection cycle. Combat sports gyms run shared mats, shared headgear, shared towels, shared everything. When one fighter brings in a stomach bug or a respiratory thing, the team gets it within days. Most fighters power through, sometimes with the help of antibiotics that aren't indicated (it's almost always viral), which restarts the antibiotic-disruption loop above. The play is to not be the one running on a compromised gut when the bug goes around, because you'll be the one who gets it worst. The same fiber, fermented foods, sleep, and stress management story that protects gut diversity also protects you in the gym when something is going through it. Probiotic supplementation in the weeks leading up to and during competition has decent randomized evidence for reducing the rate and severity of GI and respiratory infections in athletes (29% reduction in respiratory infections in one marathon study, similar or better effects in fight-camp populations in smaller trials). Probiotics during camp are one of the few supplements that earn their place as a hedge.
Add the operator-level wreckers from the previous section to this list (the chronic NSAIDs, the bad sleep, the chronic stress) and you have the full picture. Combat athletes aren't ignoring their bodies. They're putting their bodies through the meat grinder in a way that has no real civilian analog, and the gut takes the brunt of it.
The relief side is the same as for the operator, with extra weight on a few specific moves. Treat the gut hard between camps. Use the off-block to rebuild diversity, not only to "rest." Eat more plants than the camp diet allows when you're outside camp. Stack kefir, kiwi, and fermented foods on a daily basis. Be more disciplined with antibiotics than your gym is. Hydrate harder around the sauna work. These changes don't replace any of the training. They make the training pay you back.
The reset
Most of the rebuild moves are already in this piece. The food work especially. Here's the consolidated version, plus a few non-food levers we haven't unpacked yet.
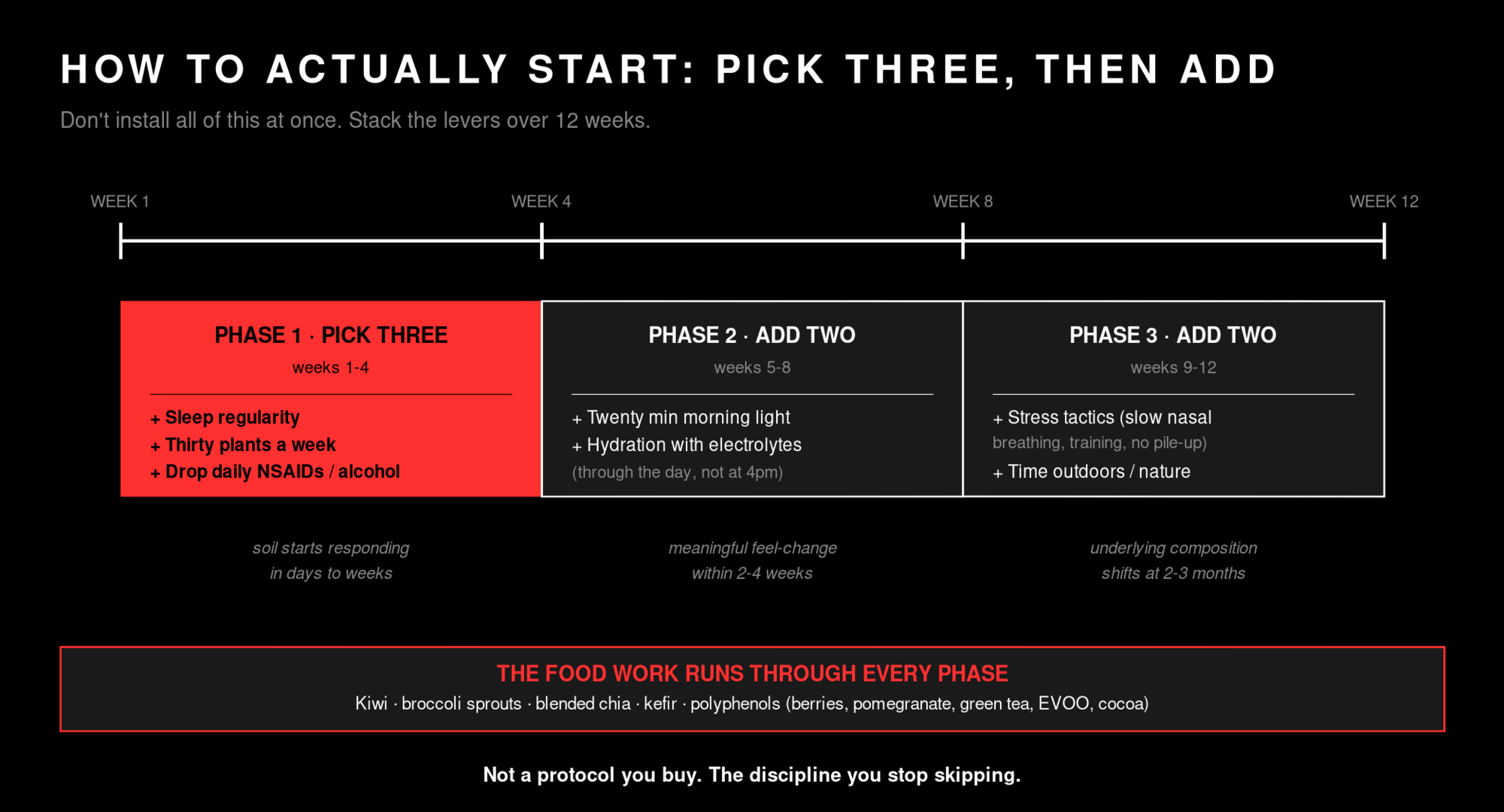
The food playbook, in one place. Aim for thirty different plants a week. Track it for two weeks on a notes app and you'll see where you actually are (most people start in the 8-15 range). Add a kiwi a day. Add a half-cup of dark berries. Add a tablespoon of blended chia in a smoothie. Add a glass of kefir on most days, especially in the morning. Cook with extra herbs and spices on whatever you're already making (oregano, thyme, rosemary, turmeric, ginger, garlic each count toward your thirty). Eat broccoli sprouts a few times a week for sulforaphane. Cycle in polyphenol-rich foods (pomegranate, green tea, extra virgin olive oil, cocoa) to feed Akkermansia and keep the mucus layer thick. Pull alcohol back to one or two drinks a week. Pull daily NSAIDs out of the rotation entirely unless there's a real injury. Don't skip protein. Don't go vegan-or-bust. Add diversity to what you're already doing.
These are the non-food rebuilders that matter most.
Sleep regularity. Duration matters, but the regularity of your sleep window matters at least as much for the microbiome. Going to bed and waking up within a 30-minute window 6-7 nights a week resets the gut clock. A 5-hour Tuesday and a 9-hour Saturday do not average out at the bacterial level. Pick a window. Defend it. If only one thing on this list changes, this one.
Morning sunlight. Twenty minutes of outdoor light within an hour of waking (no sunglasses for those twenty minutes) anchors your circadian rhythm and dampens the cortisol spike that follows you through the day. The gut clock follows your light exposure as much as it follows your food timing. This is unglamorous and free, and most operators skip it. Stop skipping it.
Hydration done well. Not "drink more water." Drink consistent water with electrolytes spread across the day. The most-common athlete pattern is dehydrated by 11am, slamming a liter at 4pm, then waking up at 2am to pee. That cycle does nothing good for the gut. A pint of water with a pinch of salt before training, sip water through the day, finish hydrating by dinner. For combat athletes between sauna sessions, the recovery window is non-negotiable.
Antibiotic and NSAID discipline. Use both when they're actually indicated. Push back on doctors who default to a Z-Pak for a viral cough or hand out NSAIDs for chronic tightness without a structural reason. After any necessary antibiotic course, rebuild aggressively for eight weeks: extra fermented foods, extra plant variety, extra sleep. This isn't paranoia. This is being the operator about your own medical inputs.
Stress practical moves. "Stress less" is useless. The actual moves that dampen the cortisol load on your gut are: morning light (already covered), exercise on most days (which you're doing), three to five minutes of slow nasal breathing once or twice a day, and not letting your high-stress weeks become your high-alcohol weeks. People who lift hard already have one of the most effective stress regulators in their week. The job is not to undo it with the rest of how you live.
Time outdoors and environmental exposure. Beyond morning light, time spent outside in actual nature (parks, hikes, gardens, the back yard) exposes you to a wider environmental microbiome than the inside of an office or a gym. People who garden have measurably more diverse gut microbiomes than people who don't, controlling for diet. You don't have to take up gardening. You do have to get yourself outdoors more than a phone call from the patio.
Movement, already named. The training you're doing is part of the rebuild. Cardiorespiratory fitness drives gut diversity. Resistance training tightens the gut wall and seeds butyrate producers. Keep doing it. Some readers reading this need a deload more than a rebuild. If you've been training through accumulating injuries, bad sleep, bad food, and chronic stress, the answer is sometimes less work, better recovery, and the soil catches up.
A note on how to start. Don't try to install all of this at once. Pick three. The highest-leverage three for most people are sleep regularity, thirty plants a week, and dropping daily NSAIDs and alcohol. Do those three for four weeks. Then add two more (morning light, hydration). Then add the last two (stress tactics, outdoor time). The soil moves faster than most people expect once the obvious wreckers retreat and the obvious inputs come back online.
This is the rebuild. It isn't a protocol you buy. It isn't a stack you order. It's the operating layer underneath everything else you're trying to do.
The other half of skin defense (for the people on mats)
If you spend time on shared mats, in shared headgear, in wrestling rooms, at jiu-jitsu gyms, in climbing facilities, or anywhere a lot of bodies sweat into the same surfaces, you need the second half of the skin defense story.
We covered the chemical half earlier: a gut-trained immune system producing antimicrobial peptides like cathelicidin that kill Staphylococcus aureus directly. That defense layer lives in your skin cells and runs on gut signals. The biological half is different. It lives on your skin's surface, and it's a population of beneficial bacteria competing for the same real estate as the pathogens that want to colonize you.
Here's the mechanism, in plain English. Your skin is not sterile. It's covered with a layer of resident bacteria, dominated by a species called Staphylococcus epidermidis. The name sounds alarming because it has "Staphylococcus" in it, but S. epidermidis is the friendly cousin of S. aureus and one of the main reasons you don't constantly catch staph infections. It deploys three kinds of weapons against S. aureus:
Quorum-sensing interference. S. aureus uses a chemical signaling system to coordinate when to form colonies, when to release toxins, and when to invade tissue. S. epidermidis releases a molecule (called AIP-I) that scrambles that signal. S. aureus shows up, can't communicate with itself, and stays inert.
Bacteriocins. S. epidermidis manufactures small protein weapons (epidermicin, nukacin, epilancin) that directly kill S. aureus cells on contact.
Real estate competition. S. epidermidis occupies the niches on your skin (hair follicles, sweat duct openings, surface oils) that S. aureus needs to colonize. A skin already crowded with friendly bacteria has nowhere for the pathogen to set up shop.
When the skin microbiome is healthy, you can roll on a mat that has staph on it and not get staph. The exposure happens; the colonization doesn't. The friendly bacteria intercept the pathogen before it gets a foothold.
The skin microbiome breaks down in two ways. Both are common in combat sports.
Over-cleaning. Daily antibacterial soap, scalding showers, hard scrubs, alcohol-based hand sanitizer all over the body. These strip the friendly bacteria along with the dirt. The opportunists come back faster than the commensals do, because S. aureus is hardier and faster-growing than S. epidermidis. The result is that the most aggressive hygiene actually leaves you more vulnerable to colonization, not less. Use plain soap (not antibacterial) on parts of your body that need it. Use warm water, not scalding. Let your skin recover its oils between sessions.
Occlusion. Wet, warm, sweaty fabric pressed against skin for hours is a culture medium. The gi you sat in after class. The rashguard that didn't get rinsed. The wrestling singlet packed wet. The headgear stored in a closed bag. Each of those creates the temperature and humidity conditions S. aureus loves. Get out of training gear within thirty minutes of finishing. Rinse off. Dry fully before clothes go back on. Wash gear after every session. This isn't optional in shared-mat sports. It's the minimum.
The numbers on shared-surface contamination are worth knowing. In one study swabbing 288 surfaces across 16 fitness facilities in Northeast Ohio, 38% tested positive for S. aureus, with the highest rates of methicillin-resistant strains (MRSA) appearing in community-associated facilities (above 50%). A separate study at a Division I university found that 76% of collegiate wrestlers carried MRSA, the highest carrier rate among the nine sports they sampled. A meta-analysis of asymptomatic colonized athletes found that carrying MRSA was associated with a sevenfold higher rate of subsequent MRSA infection.
The honest call. The gut work in this piece supports the chemical half of skin defense. It does not replace the biological half. A grappler with a clean gut, a healthy AMP response, and good mat hygiene is well-protected. A grappler with a clean gut and bad mat hygiene will still get staph occasionally. A grappler with bad gut soil and bad mat hygiene is in the most vulnerable position the sport has to offer, and most of the chronic skin issues in any gym are coming out of that group.
Practical moves that work with the gut work:
Shower within thirty minutes of training, every time. Use plain soap on the areas that contact mats (groin, armpits, feet, anywhere with abrasions). Let everywhere else just get rinsed. Don't sit in wet gear. Wash gi, rashguard, and gym clothes after every single session. Wash headgear regularly with appropriate cleaners. Treat any cut, scratch, or abrasion same-day with a barrier (Tegaderm, a cover, anything that keeps mat surface off open skin). If something on your skin looks like staph or starts spreading fast, get to a doctor. That's the moment antibiotics earn their place. Don't try to handle a real infection with topicals alone.
The defenses work in layers. Your gut feeds your skin's chemistry. Your skin's bacteria fight the pathogens directly. Your hygiene reduces the exposure. Your sleep, food, and training keep all three systems supplied. Stack the layers and you can train for years without the chronic skin issues most gyms accept as normal. Skip any of them and the gym's bug count starts landing on you.
The bill the gut keeps for you
You can pay now in boring inputs, or pay later in compounding interest.
Pay now: thirty plants a week. Water through the day with electrolytes. Twenty minutes of morning light. A regular sleep window. A glass of kefir. Two kiwis. A handful of broccoli sprouts. A walk outside instead of a phone call from the kitchen. No daily ibuprofen unless something is actually torn. One or two drinks a week instead of one or two a night. The training you're already doing.
Pay later: the cold that keeps coming. The Monday fog you can't caffeinate out of. The breakouts on your jaw at 41. The lift that won't move. The third round that dies. The skin flare two weeks out from competition. The week you can't push because your nervous system is dragging. The camp that wrecks you. The quarter that didn't go the way it was supposed to. The supplements you ordered to fix any of this that didn't fix any of this.
The gut keeps a tab. It runs on the inputs you give it and the wreckers you don't pull. When the soil is fed, you stop noticing it. When it's not, you notice it everywhere: in your immune system, your head, your performance, your skin. The two-pound organ in your large intestine is the operating layer underneath every other piece of the system you're trying to maintain. Treat it accordingly and it stops being a tax. Ignore it and it keeps charging you, with interest.
The work isn't a protocol you buy. It's the discipline you stop skipping.
Train the soil. Everything else gets easier.
*This article is educational and not a substitute for medical advice. Consult a qualified professional before making decisions about your health.
Sources
Sender R, Fuchs S, Milo R. 2016. Revised Estimates for the Number of Human and Bacteria Cells in the Body. PLOS Biology 14(8):e1002533. (Source of the 38 trillion bacterial cell estimate and the bacteria-to-human cell ratio.)
Donohoe DR, Garge N, Zhang X, et al. 2011. The microbiome and butyrate regulate energy metabolism and autophagy in the mammalian colon. Cell Metabolism 13(5):517-526. (Butyrate as the primary energy substrate for colonocytes; the ~70% of colon-cell energy figure.)
Furusawa Y, Obata Y, Fukuda S, et al. 2013. Commensal microbe-derived butyrate induces the differentiation of colonic regulatory T cells. Nature 504(7480):446-450. (The mechanism by which butyrate drives regulatory T cell production, central to the "peacekeeper" framing.)
Bermon S, Petriz B, Kajėnienė A, Prestes J, Castell L, Franco OL. 2015. The microbiota: an exercise immunology perspective. Exercise Immunology Review 21:70-79. (Exercise-induced intestinal ischemia and transient gut barrier permeability during heavy training.)
Pedersen BK, Hoffman-Goetz L. 2000. Exercise and immune function: recent developments. Physiological Reviews 80(3):1055-1081. (Original framing of the post-exercise "open window" of immunosuppression.)
Campbell JP, Turner JE. 2018. Debunking the myth of exercise-induced immune suppression: redefining the impact of exercise on immunological health across the lifespan. Frontiers in Immunology 9:648. (The modern critique reframing the open window as immune cell redistribution rather than suppression.)
Cox AJ, Pyne DB, Saunders PU, Fricker PA. 2010. Oral administration of the probiotic Lactobacillus fermentum VRI-003 and mucosal immunity in endurance athletes. British Journal of Sports Medicine 44(4):222-226. (Probiotic supplementation in elite distance runners; approximately halved the days of respiratory symptoms vs placebo (30 days vs 72 days) over a 4-month winter training period.)
Tavares-Silva E, Caris AV, Santos SA, Ravacci GR, Thomatieli-Santos RV. 2021. Effect of Multi-Strain Probiotic Supplementation on URTI Symptoms and Cytokine Production by Monocytes after a Marathon Race: A Randomized, Double-Blind, Placebo Study. Nutrients 13(5):1478. (Marathon runners taking a multi-strain probiotic for 30 days pre-race had 29% lower incidence of URTI symptoms vs placebo in the week after the race; this is the source of the 29% URTI reduction figure.)
Kekkonen RA, Vasankari TJ, Vuorimaa T, Haahtela T, Julkunen I, Korpela R. 2007. The effect of probiotics on respiratory infections and gastrointestinal symptoms during training in marathon runners. International Journal of Sport Nutrition and Exercise Metabolism 17(4):352-363. (Lactobacillus rhamnosus GG trial in marathon runners; the 57% shorter GI symptom duration figure.)
Bonaz B, Bazin T, Pellissier S. 2018. The Vagus Nerve at the Interface of the Microbiota-Gut-Brain Axis. Frontiers in Neuroscience 12:49. (Vagus nerve as bidirectional cable between gut and brain, the ~80% afferent figure, and the four-pathway framing underlying the gut-brain chapter.)
Strandwitz P. 2018. Neurotransmitter modulation by the gut microbiota. Brain Research 1693(Pt B):128-133. (Gut bacteria as direct producers of GABA, serotonin, dopamine, and acetylcholine.)
Agudelo LZ, Femenía T, Orhan F, et al. 2014. Skeletal Muscle PGC-1α1 Modulates Kynurenine Metabolism and Mediates Resilience to Stress-Induced Depression. Cell 159(1):33-45. (Skeletal muscle as a "kynurenine sink" during exercise; the broader tryptophan-kynurenine pathway in mood regulation.)
Kennedy PJ, Cryan JF, Dinan TG, Clarke G. 2017. Kynurenine pathway metabolism and the microbiota-gut-brain axis. Neuropharmacology 112(Pt B):399-412. (Inflammation tilting tryptophan toward kynurenine and away from serotonin; the ~95/5 split.)
Stilling RM, van de Wouw M, Clarke G, Stanton C, Dinan TG, Cryan JF. 2016. The neuropharmacology of butyrate: The bread and butter of the microbiota-gut-brain axis? Neurochemistry International 99:110-132. (Butyrate crossing the blood-brain barrier and modulating microglia.)
Foster JA, Rinaman L, Cryan JF. 2017. Stress & the gut-brain axis: Regulation by the microbiome. Neurobiology of Stress 7:124-136. (HPA axis and microbiome bidirectional feedback loop.)
Scheiman J, Luber JM, Chavkin TA, et al. 2019. Meta-omics analysis of elite athletes identifies a performance-enhancing microbe that functions via lactate metabolism. Nature Medicine 25(7):1104-1109. (The Veillonella atypica marathon-runner study; lactate-to-propionate metabolism and treadmill endurance in mice. The keystone citation for the performance section.)
Petersen LM, Bautista EJ, Nguyen H, et al. 2017. Community characteristics of the gut microbiomes of competitive cyclists. Microbiome 5(1):98. (Prevotella abundance correlates with training volume in cyclists.)
Barton W, Penney NC, Cronin O, et al. 2018. The microbiome of professional athletes differs from that of more sedentary subjects in composition and particularly at the functional metabolic level. Gut 67(4):625-633. (Functional metagenomic differences between professional rugby athletes and sedentary controls.)
Clarke SF, Murphy EF, O'Sullivan O, et al. 2014. Exercise and associated dietary extremes impact on gut microbial diversity. Gut 63(12):1913-1920. (Higher microbiome diversity in elite athletes; correlation with exercise and protein intake.)
Estaki M, Pither J, Baumeister P, Little JP, Gill SK, Ghosh S, Ahmadi-Vand Z, Marsden KR, Gibson DL. 2016. Cardiorespiratory fitness as a predictor of intestinal microbial diversity and distinct metagenomic functions. Microbiome 4(1):42. (VO2peak accounts for >20% of variance in gut taxonomic richness, independent of diet.)
Allen JM, Mailing LJ, Niemiro GM, et al. 2018. Exercise alters gut microbiota composition and function in lean and obese humans. Medicine & Science in Sports & Exercise 50(4):747-757. (Six-week exercise intervention shifts microbiome composition and increases short-chain fatty acid production, independent of diet.)
Cullen JM, Shahzad S, Kanaley JA, Ericsson AC, Dhillon J. 2024. The effects of 6 wk of resistance training on the gut microbiome and cardiometabolic health in young adults with overweight and obesity. Journal of Applied Physiology 136(2):349-361. (6-week resistance training in sedentary young adults with overweight/obesity increased Roseburia abundance (an SCFA producer) and improved insulin sensitivity and diastolic blood pressure. Wider literature suggests resistance training also lowers zonulin and supports mucin production.)
Trappe TA, White F, Lambert CP, Cesar D, Hellerstein M, Evans WJ. 2002. Effect of ibuprofen and acetaminophen on postexercise muscle protein synthesis. American Journal of Physiology - Endocrinology and Metabolism 282(3):E551-E556. (NSAIDs do not reduce delayed-onset muscle soreness and may impair muscle adaptation.)
Van Wijck K, Lenaerts K, Van Bijnen AA, et al. 2012. Aggravation of exercise-induced intestinal injury by Ibuprofen in athletes. Medicine & Science in Sports & Exercise 44(12):2257-2262. (Pre-exercise ibuprofen elevates I-FABP and increases small intestinal permeability vs. exercise alone.)
Bjarnason I, Hayllar J, Macpherson AJ, Russell AS. 1993. Side effects of nonsteroidal anti-inflammatory drugs on the small and large intestine in humans. Gastroenterology 104(6):1832-1847. (All NSAIDs increase intestinal permeability within 24 hours of ingestion.)
Engen PA, Green SJ, Voigt RM, Forsyth CB, Keshavarzian A. 2015. The Gastrointestinal Microbiome: Alcohol Effects on the Composition of Intestinal Microbiota. Alcohol Research: Current Reviews 37(2):223-236. (Alcohol effects on tight junctions and gut microbial composition; loss of Lactobacillus and Bifidobacterium.)
Bishehsari F, Magno E, Swanson G, et al. 2017. Alcohol and Gut-Derived Inflammation. Alcohol Research: Current Reviews 38(2):163-171. (Alcohol-induced gut permeability and bacterial translocation producing systemic inflammation.)
Dethlefsen L, Relman DA. 2011. Incomplete recovery and individualized responses of the human distal gut microbiota to repeated antibiotic perturbation. Proceedings of the National Academy of Sciences 108 Suppl 1:4554-4561. (Single courses of antibiotics produce incomplete microbiome recovery, with some species not returning at six months.)
Thaiss CA, Zeevi D, Levy M, et al. 2014. Transkingdom control of microbiota diurnal oscillations promotes metabolic homeostasis. Cell 159(3):514-529. (Circadian disruption and microbiome diurnal rhythms; jet lag and shift work effects on bacterial composition.)
Voigt RM, Forsyth CB, Green SJ, et al. 2014. Circadian disorganization alters intestinal microbiota. PLOS ONE 9(5):e97500. (Circadian disruption reduces beneficial species including Akkermansia muciniphila.)
Smith RP, Easson C, Lyle SM, et al. 2019. Gut microbiome diversity is associated with sleep physiology in humans. PLOS ONE 14(10):e0222394. (Sleep quality, regularity, and microbiome diversity associations.)
McDonald D, Hyde E, Debelius JW, et al. 2018. American Gut: An Open Platform for Citizen Science Microbiome Research. mSystems 3(3):e00031-18. (Origin of the 30-plants-per-week metric; plant diversity over total fiber as predictor of microbiome richness.)
Wastyk HC, Fragiadakis GK, Perelman D, et al. 2021. Gut-microbiota-targeted diets modulate human immune status. Cell 184(16):4137-4153.e14. (Stanford fermented foods randomized trial: 10-week intervention increased microbiome diversity and reduced 19 inflammatory markers including IL-6.)
Anhê FF, Roy D, Pilon G, et al. 2015. A polyphenol-rich cranberry extract protects from diet-induced obesity, insulin resistance and intestinal inflammation in association with increased Akkermansia spp. population in the gut microbiota of mice. Gut 64(6):872-883. (Polyphenols increase Akkermansia muciniphila abundance.)
Roopchand DE, Carmody RN, Kuhn P, et al. 2015. Dietary polyphenols promote growth of the gut bacterium Akkermansia muciniphila and attenuate high-fat diet-induced metabolic syndrome. Diabetes 64(8):2847-2858. (Mechanistic study of the polyphenol-Akkermansia axis.)
Salem I, Ramser A, Isham N, Ghannoum MA. 2018. The gut microbiome as a major regulator of the gut-skin axis. Frontiers in Microbiology 9:1459. (Comprehensive review of the gut-skin axis: how dysbiosis drives inflammatory skin conditions through systemic immune modulation.)
De Pessemier B, Grine L, Debaere M, Maes A, Paetzold B, Callewaert C. 2021. Gut-Skin Axis: Current Knowledge of the Interrelationship between Microbial Dysbiosis and Skin Conditions. Microorganisms 9(2):353. (Reviews atopic dermatitis, acne, and psoriasis as downstream manifestations of gut dysbiosis; integrates Th2/Th17 immune mechanisms.)
Vaughn AR, Notay M, Clark AK, Sivamani RK. 2017. Skin-gut axis: The relationship between intestinal bacteria and skin health. World Journal of Dermatology 6(4):52-58. (Mechanistic review of intestinal bacteria and skin inflammation pathways.)
Wang Z, Klipfell E, Bennett BJ, et al. 2011. Gut flora metabolism of phosphatidylcholine promotes cardiovascular disease. Nature 472(7341):57-63. (Foundational paper from the Hazen lab establishing TMAO as a gut-derived driver of atherosclerotic cardiovascular disease.)
Tang WH, Wang Z, Levison BS, et al. 2013. Intestinal microbial metabolism of phosphatidylcholine and cardiovascular risk. New England Journal of Medicine 368(17):1575-1584. (3-year follow-up of 4,007 patients undergoing coronary angiography; elevated plasma TMAO independently predicted heart attack, stroke, and death.)
Multi-Ethnic Study of Atherosclerosis investigators. 2025. Trimethylamine-N-oxide (TMAO) and risk of incident cardiovascular events in the multi-ethnic study of Atherosclerosis. Scientific Reports. DOI: 10.1038/s41598-025-05903-3. (Highest TMAO quintile had ~33% higher hazard of atherosclerotic cardiovascular events vs. lowest quintile, with graded risk across quintiles.)
Heianza Y, Ma W, DiDonato JA, et al. 2020. Long-term changes in gut microbial metabolite trimethylamine-N-oxide and coronary heart disease risk. Journal of the American College of Cardiology 75(7):763-772. (Changes in TMAO over time predict coronary heart disease independent of baseline; supports the modifiability argument.)
Yang T, Santisteban MM, Rodriguez V, et al. 2015. Gut dysbiosis is linked to hypertension. Hypertension 65(6):1331-1340. (Distinct microbial composition in hypertensive populations vs. normotensive; reduced SCFA-producing species in hypertension.)
Pluznick JL. 2017. Microbial short-chain fatty acids and blood pressure regulation. Current Hypertension Reports 19(4):25. (Mechanism: SCFA receptors (Olfr78, Gpr41, Gpr43) on vascular endothelium mediate blood pressure effects of gut-derived SCFAs.)
Verhaar BJH, Prodan A, Nieuwdorp M, Muller M. 2020. Gut microbiota in hypertension and atherosclerosis: A review. Nutrients 12(10):2982. (Synthesis of the gut-cardiovascular axis evidence; integration of TMAO and SCFA pathways.)
Jama HA, Beale A, Shihata WA, Marques FZ. 2019. The effect of diet on hypertensive pathology: is there a link via gut microbiota-driven immunometabolism? Cardiovascular Research 115(9):1435-1447. (Dietary fiber as a lever for SCFA-driven cardiovascular protection.)
Battson ML, Lee DM, Weir TL, Gentile CL. 2018. The gut microbiota as a novel regulator of cardiovascular function and disease. Journal of Nutritional Biochemistry 56:1-15. (Comprehensive review of gut microbiota effects on endothelial function, blood pressure, and atherosclerosis.)
Ong PY, Ohtake T, Brandt C, et al. 2002. Endogenous antimicrobial peptides and skin infections in atopic dermatitis. New England Journal of Medicine 347(15):1151-1160. (Patients with atopic dermatitis have measurably lower cathelicidin (LL-37) and beta-defensin levels in their skin, correlated with elevated S. aureus colonization rates.)
Eyerich K, Pennino D, Scarponi C, et al. 2009. IL-17 in atopic eczema: linking allergen-specific adaptive and microbial-triggered innate immune response. Journal of Allergy and Clinical Immunology 123(1):59-66. (Mechanism: IL-4 and IL-13 antagonize Th17-driven antimicrobial peptide production in keratinocytes.)
Parodi A, Paolino S, Greco A, et al. 2008. Small intestinal bacterial overgrowth in rosacea: clinical effectiveness of its eradication. Clinical Gastroenterology and Hepatology 6(7):759-764. (Documented overlap between SIBO and rosacea; treatment of SIBO produces substantial rosacea improvement.)
Ricci AA, Evans C, Stull C, Peacock CA, French DN, Stout JR, Fukuda DH, La Bounty P, Kalman D, Galpin AJ, Tartar J, Johnson S, Kreider RB, Kerksick CM, Campbell BI, Jeffery A, Algieri C, Antonio J. 2025. International Society of Sports Nutrition Position Stand: nutrition and weight cut strategies for mixed martial arts and other combat sports. Journal of the International Society of Sports Nutrition 22(1):2467909. (Combat sports weight-cut prevalence (60-80%), gradual cut recommendations, dehydration as last lever, post-weigh-in recovery.)
Otto M. 2009. Staphylococcus epidermidis – the 'accidental' pathogen. Nature Reviews Microbiology 7(8):555-567. (S. epidermidis as commensal counterpart to S. aureus, including the bacteriocin and quorum-sensing-interference defense mechanisms.)
Iwase T, Uehara Y, Shinji H, et al. 2010. Staphylococcus epidermidis Esp inhibits Staphylococcus aureus biofilm formation and nasal colonization. Nature 465(7296):346-349. (Direct interspecies inhibition mechanism in skin commensal-pathogen competition.)
Dalman M, Bhatta S, Nagajothi N, Thapaliya D, Olson H, Naimi HM, Bhatta DR, Smith TC. 2019. Characterizing the molecular epidemiology of Staphylococcus aureus across and within fitness facility types. BMC Infectious Diseases 19:69. DOI: 10.1186/s12879-019-3699-7. (288 environmental swabs across 16 fitness facilities in Northeast Ohio; 38.2% positive for S. aureus, with higher MRSA contamination in community-associated facilities (52.8%) than in hospital-associated (5.56%) or CrossFit (14.3%) facilities.)
Champion AE, Goodwin TA, Brolinson PG, Werre SR, Prater MR, Inzana TJ. 2014. Prevalence and characterization of methicillin-resistant Staphylococcus aureus isolates from healthy university student athletes. Annals of Clinical Microbiology and Antimicrobials 13:33. (76% of collegiate wrestlers at a Division I university carried MRSA, the highest carriage rate among nine sports sampled; adjusted odds ratio 29.7 for wrestling.)
Karanika S, Kinamon T, Grigoras C, Mylonakis E. 2016. Colonization With Methicillin-resistant Staphylococcus aureus and Risk for Infection Among Asymptomatic Athletes: A Systematic Review and Metaanalysis. Clinical Infectious Diseases 63(2):195-204. (Meta-analysis of asymptomatic athlete cohorts; MRSA colonization associated with a sevenfold higher rate of subsequent MRSA skin/soft tissue infection within 3 months. Pooled prevalence of MRSA colonization in athletes was 6%, higher in US athletes (8%) and collegiate athletes (13%), with wrestling at 22%.)